 
Power cycles are cycles that convert some heat input into mechanical work output. The thermodynamic cycles can be divided into two primary classes: It is the most important law for analyzing most systems and quantifying how thermal energy is transformed to other forms of energy. It is the principle of conservation of energy, meaning that energy can neither be created nor destroyed but rather transformed into various forms as the fluid within the control volume is being studied. The increase in internal energy of a closed system is equal to the heat supplied to the system minus the work done by it. The first law of thermodynamics dictates that the net heat input equals the network output over any cycle. A typical thermodynamic cycle consists of a series of thermodynamic processes transferring heat and work while varying pressure, temperature, and other state variables, eventually returning a system to its initial state. At the conclusion of a cycle, all the properties have the same value they had initially. Many heat engines operate in a cyclic manner, adding energy in the form of heat in one part of the cycle and using that energy to do useful work in another part of the cycle.Ī process that eventually returns a system to its initial state is called a cyclic process. A heat engine must be used to convert thermal energy into another form of energy. Therefore these energy sources are so-called “ primary energy sources” that must be converted to the secondary energy source, so-called energy carriers ( electrical energy, etc.). But the burning of fossil fuels generates only thermal energy. 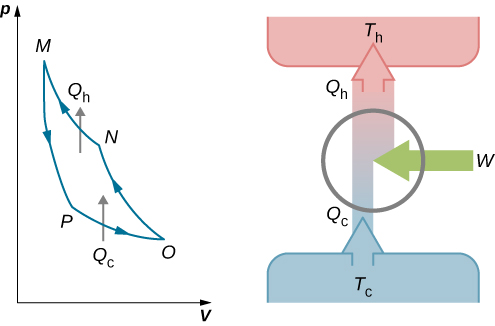
At present, fossil fuel is still the world’s predominant energy source. Our goal here will be to introduce thermodynamics as the energy conversion science. In general, thermodynamics is the science that deals with energy production, storage, transfer, and conversion. Today, the Rankine cycle is the fundamental operating cycle of all thermal power plants where an operating fluid is continuously evaporated and condensed. Typical thermodynamic cycle consists of a series of thermodynamic processes transferring heat and work while varying pressure, temperature, and other state variables, eventually returning a system to its initial state. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
